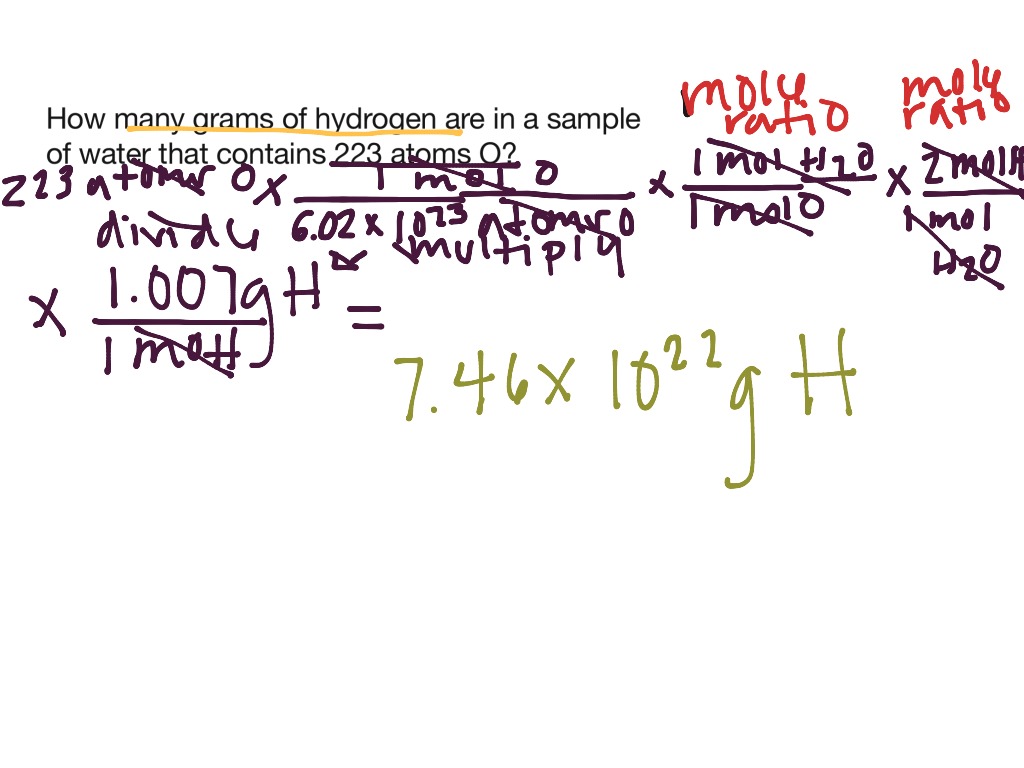
To determine the number of atoms in 1 g of the element, students should remember to multiply the value by 2. The free online mole calculator is an efficient way to find the number of moles in a substance. This is because in both cases, 1 molecule is made of 2 atoms. Hence, for these 2 elements, students should multiply the number of particles (which is molecules) by 2 to determine the number of atoms. Here, one of the elements, iron, is made of atoms, while the other two, iodine and oxygen, are made of molecules. In this video, we will focus on determining the number of atoms in 3 different elements of the same mass. Midpoint calculator is a simple to use and very easy app to find the mid-value of two points without hassle. Hence, we can safely say that, to determine the number of particles, we can use this formula: number of particles = number of moles x 6 x 10 23 In this Video Since 1 mole of substance contains 6 x 10 23 particles, 2 moles of substance contains 2 x 6 x 10 23 particles.0.5 moles of substance contains 0.5 x 6 x 10 23 particles. The formula for converting molecules to moles is a little different than mass and volume because both molecules and moles are measures of quantity: mol molecules 6.02214076 × 10 23 The quantity of substance in moles is equal to the number of molecules divided by the Avogadro constant (6.02214076 × 10 23 ). Formula: number of particles = number of moles x 6 x 10 23 The tool will immediately convert the moles to grams and calculate atoms present in it. The value 6 x 10 23 is called Avogadro’s constant or Avogadro’s number.Įqual number of moles of substances contain the same number of particles. Moles To Grams Calculator Select the chemical from the list or simply enter the molar mass of your choice. In 1 mol of hydrogen gas, 6 x 10 23 molecules have 2 x 6 x 10 23 hydrogen atoms. 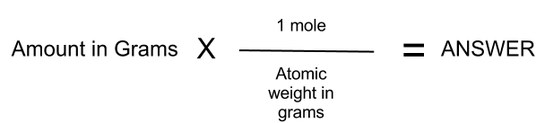
This is because 1 hydrogen molecule has 2 atoms. Molar mass calculator also displays common compound name, Hill formula. To determine its number of atoms, we need to multiply the number of molecules by 2. Enter a chemical formula to calculate its molar mass and elemental composition. If we focus on hydrogen gas, then 1 mol of hydrogen gas has 6 x 10 23 hydrogen molecules. A mole of a substance or a mole of particles is defined as exactly 6.02214076×10²³ particles, which may be atoms, molecules, ions, or electrons. The particles can be atoms, molecules, ions or even electrons, depending on what substance we are referring to.įor example, if we focus on helium, then we are talking about helium atoms. One mole of a substance has 6 x 10 23 particles. mole-mass calculation, where we start with a given number of moles of a substance and calculate the mass of another substance involved in the chemical. Hence, chemist use a unit – mole – to count particles. There are so many of them it is pretty impossible to count them. This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. Round your answer to 3 significant digits. Also, they must be confident with calculating the number of particles from the number of moles of particles and vice versa. Calculate the millimoles of calcium sulfate the chemist has added to the flask. Iodine (I 2) iron (Fe) oxygen (O 2) Syllabus ObjectivesĪt O levels, students must be able to perform calculations involving the number of moles, mass and molar mass of a substance. Then the coefficients of the balanced chemical reaction can be used to convert to moles of another substance, which can then be converted to a mass.įor example, let us determine the number of grams of SO 3 that can be produced by the reaction of 45.Which substance contains the greatest number of atoms in 1 g? provides an online conversion calculator for all. Again, however, it is important to emphasize that before the balanced chemical reaction is used, the mass quantity must first be converted to moles. This is the number of molecules in 1 mole of a chemical compound. The formula looks like this: moles grams of compound/molar mass of compound 2 Plug your numbers into the formula. For this type of calculation, the molar masses of two different substances must be used-be sure to keep track of which is which. The number of moles you have of a compound can be calculated by dividing the number of grams of the compound by the molecular mass of the compound. It should be a trivial task now to extend the calculations to mass-mass calculations, in which we start with a mass of some substance and end with the mass of another substance in the chemical reaction. 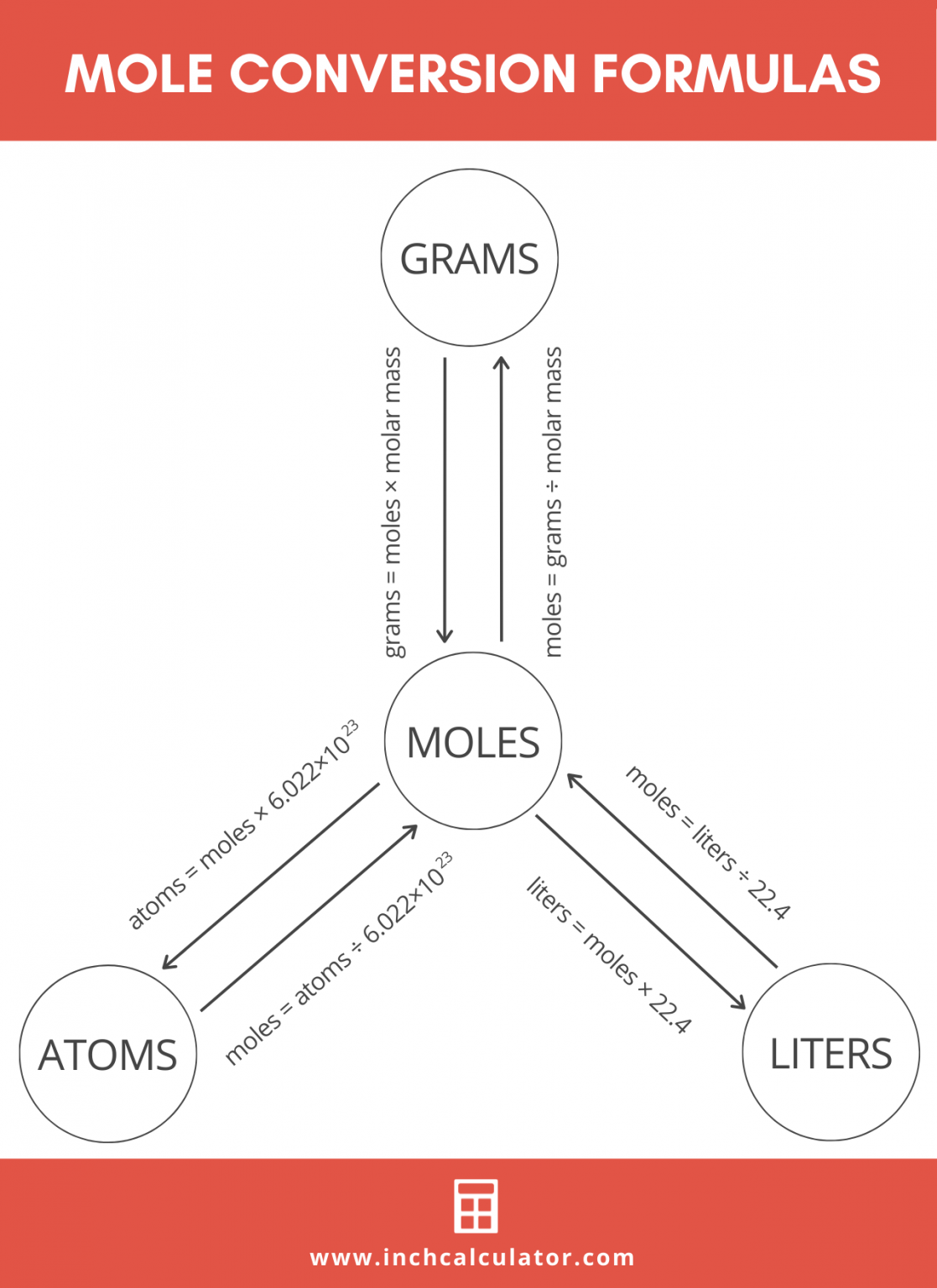
This module shows how the mole, known as Avogadro’s number, is key to calculating quantities of atoms and molecules. \nonumber \] Answerģ0.4 g (Note: here we go from a product to a reactant, showing that mole-mass problems can begin and end with any substance in the chemical equation.) The mole is an important concept for talking about a very large number of things 6.02 x 10 23 of them to be exact.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
